Technology
Technology - Innovation Driving Rapid Growth
PATENTED TECHNOLOGIES
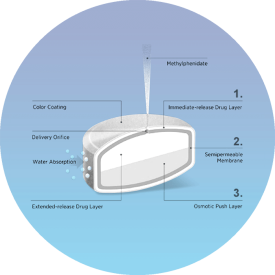
TIME-RELEASED TABLETS
- Up to 24 hours sustained release of active compounds in the gastrointestinal tract.
- Time-specific release of active compounds to achieve a wide set of targeted effects.
- Incorporation of up to 600 mg of API on 1500 mg embossed tablet.

BUCCAL TABLETS
- Dissolves between the gums & cheek through the oral mucosa.
- Potent drugs are slowly absorbed into the blood for extended effect.
- Lasts up to 30min.

SUBLINGUAL TABLETS
- Rapid Onset.
- Disintegrates in seconds above or below the tongue.
- Inexpensive formulation and manufacturing process.
- Ability to incorporate modified release components for controlled release of the API according to a desired PK profile.
COMPLIMENTARY TECHNOLOGIES
REGULAR TABLETS
- Easy to swallow.
- No measurement required.
- More stable than other dosage forms.
- Low-cost.

GRANULAR IN SACHETS
- Granular preparations available in effervescent or non-effervescent form (sachets).
- More stable than powder-based preparations.
- Moisture protection.
- Easy storage.

EFFERVESCENT PRODUCTS
- Effervescence generated by an autocatalytic reaction catalysed by water and generating CO2 in the solution.
- Reaction is modulated by strict control of residual moisture.
- Optimal compatibility for stomach absorbance.
- Maintains bio-equivalence.
- Taste masking & perfect dissolution.
MANUFACTURING
Our manufacturing facilities produce more than 200 million units annually. We produce pharmaceutical products in a dedicated area that is independent and completely separated from the nutraceutical production area. Alongside our other facilities, our manufacturing plants systematically qualify for routine Good Practice inspections.

CORE MANUFACTURING CAPABLITIES
- Loading
- Granulation
- Cleaning stations
- Blending and unloading
- Tableting
- Tubbing
- Water purification Tanks
Warehouse
No lab is truly complete without a PGW (pharma-grade warehouse). Our FDA- and Swissmedic-certified warehouse provides industry-leading reliability for storing sensitive medicines and products, achieved through constant monitoring of utilities that could impact the products’ quality such as humidity and vapour, gases, heat, and conventional air conditioning.

OUR WAREHOUSE FEATURES
- 1,000m² with 1,200 pallet capacity
- Humidity control and moisture protection
- Handling of narcotics and controlled substances
- Global logistics and export facility
- State-of-the-art security and monitoring equipment
- Next door to the Alpex laboratory and manufacturing facility

